BDP FL Maleimide - http://www.lumiprobe.com/p/bodipy-fl-maleimide  BDP FL - borondipyrromethene dye is an excellent dye for fluorescein (FAM) channel. BDP FL - borondipyrromethene dye is an excellent dye for fluorescein (FAM) channel.Thiol reactive BDP FL maleimide is a reactive dye for protein labeling, which has identical structure with BODIPY ® FL maleimide. BDP FL is a borondipyrromethene dye which has absorption and fluorescence spectra similar to fluorescein (FAM). However, this dye exhibits very high photostability. It is non-charged, and has low molecular weight. Its brightness is similar to fluorescein, R110 and xanthene dye derivatives like Alexa Fluor® 488. This fluorophore is ideal for fluorescent microscopy and many other applications. The fluorophore can substitute fluorescein for almost any application, and it is compatible with any FAM-capable fluorescent instrumentation. |
 BDP FL azide http://www.lumiprobe.com/p/bodipy-fl-azide BDP FL azide http://www.lumiprobe.com/p/bodipy-fl-azideBDP FL azide is an analog of BODIPY ® FL azide, a Click-chemistry capable bright and photostable dye for FAM channel. This green-emitting fluorophore is compatible with all types of fluorescence measuring instruments for FAM (fluorescein) and dyes like Alexa® Fluor 488. The fluorophore is a representative of borondipyrromethene class of fluorescent dyes, which possess high quantum yields in aqueous environments, and high stability towards photobleaching.  BDP FL NHS ester http://www.lumiprobe.com/p/bodipy-fl-nhs-ester BDP FL NHS ester http://www.lumiprobe.com/p/bodipy-fl-nhs-esterBDP FL NHS ester is an advanced dye for 488 nm channel, a replacement for fluorescein, a molecule identical to BODIPY FL ® NHS ester. An amino-reactive dye for the labeling of proteins and peptides. While the absorbance and emission spectra of this molecule stay within FAM excitation and emission channels, this dye provides much better photostability, and outstanding brightness. The fluorescence spectrum of BODIPY-FL is narrower than that of FAM. This provides a better brightness for monochromator based instruments, when emission wavelength can be tuned to dye maximum. The dye is neutral, possesses low molecular weight, and retain high quantum yield in conjugates. The dye is a good replacement for fluorescein (FAM), BODIPY-FL, Alexa Fluor 488, DyLight 488, Cy2, and other 488 nm dyes. Citations of interest - Take a look! Ultrasensitive fluorescence-based methods for nucleic acid detection: towards amplification-free genetic analysis 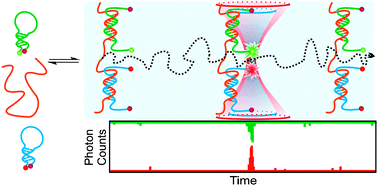 Ranasinghe, T.; Brown, T. Chem. Commun., 2011,47, 3717-3735 DOI: 10.1039/C0CC04215C Real time PCR is the mainstay of current nucleic acid assays, underpinning applications in forensic science, point-of-care diagnostics and detection of bioterrorism agents. Despite its broad utility, the search for new tests continues, inspired by second and third generation DNA sequencing technologies and fuelled by progress in single molecule fluorescence spectroscopy, nanotechnology and microfabrication. These new methods promise the direct detection of nucleic acids without the need for enzymatic amplification. In this feature article, we provide a chemist's perspective on this multidisciplinary area, introducing the concepts of single molecule detection then focussing on the selection of labels and probe chemistry suitable for generating a signal detectable by ultrasensitive fluorescence spectroscopy. Finally, we discuss the further developments that are required to incorporate these detection platforms into integrated ‘sample-in-answer-out’ instruments, capable of detecting many target sequences in a matter of minutes. |
Tech Support: Lumiprobe offers FREE tech support before or after you order. A few questions: I just ordered the BDP FL-azide dye that I want to click onto my peptides which have an alkyne. Do you have a protocol for running this click reaction in solution? The one which you provide on the website is for oligonucleotides and I was wondering if you have a protocol more specific to peptides? Lumiprobe recommends you to start with our oligonucleotide protocol. Compared to oligos, peptides have a couple of peculiarities. First, they may contain thiol groups which adversely affect Cu-catalyzed reaction. If this is your case and conjugation does not perform well, try increasing Cu catalyst concentration, and do your best to exclude oxygen from the reaction. If there are no thiols, conjugation is normally non- problematic. Second, purification of the peptides is different - for example, acetone precipitation may not be appropriate. Use your favorite purification method, such as HPLC. The protocol for oligonucleotides is appropriate for peptides in all other aspects. I am looking for a probe to use in a polarization study of protein association-dissociation. The protein is a dimer of 100 kD that dissociates into two equal 50 kD monomers, so the change in polarization is very small using fluorophores with lifetimes like fluorescein or rhodamine, i.e. few nsec. The Kd for the protein dissociation is low, 50 nM, so the fluorophore needs to be pretty bright also. So, I'm looking for the usually incompatible combination of : a) high extinction coefficient, b) high quantum yield, and c) longer lifetime. It would be nice if the absorbance max is longer wavelength than fluorescein, but in any case must be longer than 350 nm. I would prefer NHS or maleimide chemistry, but can work with other options also. Thank you for your email! It is quite a difficult task to find a dye fitting all of the desired properties. There are no dyes of Lumiprobe range that fulfill all of them, and I do not know about such products among competing product ranges. BDP FL is a fluorescein-like dye which has similar brightness (i.e. high extinction coefficient and quantum yield), and reported to have fluorescence lifetime about 50% higher than fluorescein. Pyrene is a polyaromatic hydrocarbon dye which is reported to have much longer fluorescence lifetime (about 25-fold of fluorescein). However, extinction coefficient is not high (about 1/2 of fluorescein), and it is a blue emitter (emission max around 450 nm). Lumiprobe offers Pyrene azide 1 and Pyrene azide 2 and also BDP FL NHS esters. The pyrene azide dyes also exhibits effect of excimer formation (you can consider this effect to study association, too). Among Lumiprobe's cyanine dyes, there are red and NIR fluorophores, but they have lower quantum yields and fluorescence lifetimes similar to fluorescein, although their extinction coefficients are much higher. So although I cannot suggest the product that fits all requirements, hopefully you can tailor some available products to your task! I’d like to order azide dye for click reaction and like to know if you have green colour as I see only cy3 and cy5 in your website. If you do have green colour azide, could you please also send the quotation? Lumiprobe can offer FAM azides - FAM azide 6-isomer and FAM azide 5-isomer , the two isomers have very similar properties and also BDP FL azide (an analog of BODIPY FL). Both are for FAM green channel. Thanks a lot for your information. I am wondering if this FAM azide will be suitable for flow cytometry application as I have a plan to do both microscopy and FACS. FAM is suitable for flow cytometry and FACS if your instrument supports this channel (almost all of them do). BDP FL is also another dye for this channel, it is more photostable. I'm searching for a protocol that would allow me to attach a fluorescent protein to a non-fluorescent one, possibly using the click chemistry. I was wondering if this is at all possible and if so, how many modifications/protein there are on average. I'm dealing with an enzyme and I care about preserving its catalytic activity hence I was wondering if it's possible to control the extent of its modification in order to limit it as much as possible. Preferably I'd like to have just few alkyne groups/protein. It is possible to make conjugates between different proteins via Click Chemistry. You can label one with azide NHS ester, and another with alkyne activated ester , and then click them together. However, there are more straightforward ways to achieve labeling: If you need to label a protein, you can just use one of Lumiprobe's dye activated esters . For example, BDP-FL label will fit GFP fluorescent channel, other fluorescent proteins can be covered by other dyes, too. This will more likely retain your catalytic activity, because fluorescent dyes are small, and likely will label somewhere away from your catalytic site. If you definitely need to label with fluorescent protein, it is probably better to use genetic engineering to produce a chimeric protein. Please do not hesitate to contact Lumiprobe if you have more questions! Do you would suggest to label the enzyme directly with a dye without using the click chemistry. Do you know how many dyes per enzyme one usually gets using this approach? Does your company have an experience of labelling enzymes? Thank you! It is possible to make different loadings of dye per protein molecule with this approach. Lumiprobe has experience in labeling proteins, but enzymes should not be very different if they tolerate pH around 8. We recommend you to use sulfonated dyes like sulfo-Cyanine3 NHS ester and sulfo-Cyanine5 NHS ester to label enzymes specifically in very mild conditions. Although the reaction looks simple, nevertheless, all substrates are different. In some reactions, it is possible that there is some chemical reason why labeling does not take place. |
Visit Lumiprobe.com
Replies